By mobilizing the right partners across our value chain and engaging our employees to move forward together
For a company like Ipsen, environmental impact doesn’t stop at the boundaries of its operations. In fact, that’s only a small part of the picture.
Most of our footprint lies elsewhere across our value chain. It is shaped by the partners we work with, the materials we source, the way our medicines are developed and delivered. Today, around 90% of our emissions fall under Scope 3, reflecting the complexity of the pharmaceutical ecosystem.
This reality is reshaping how we approach sustainability.
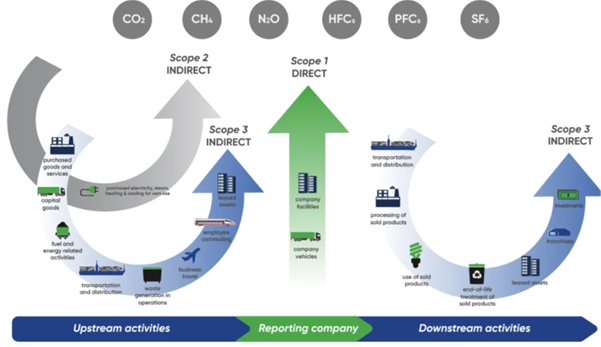
In recent years, Ipsen has made significant progress in reducing emissions from its own operations. Scope 1 and 2 emissions have been cut by more than half compared to 2019, driven by improvements in energy efficiency, electrification, and more sustainable operational choices In parallel, we have also taken action on key parts of our Scope 3 footprint, including business travel, fleet, packaging, and employee commuting, areas where change can be implemented more directly. These results demonstrate that progress is already underway, and that growth and environmental responsibility can move forward together.
The next phase is different. It is less about what we can do alone, and more about what we can do together.
We are now expanding our approach to Scope 3, moving from ambition and frameworks to execution with our suppliers and partners. Reducing these emissions requires a shift in how an entire ecosystem operates. It means partnering with our key suppliers to better understand their emissions and to identify where meaningful reductions can be achieved. It means moving beyond estimates to more precise data, enabling targeted and measurable action. And it means embedding sustainability into core business processes across our purchasing process, from strategy to operations.
In a highly regulated industry like pharmaceuticals, change does not happen overnight. Supply chains are complex, and even seemingly simple evolutions such as packaging require time, validation, and regulatory approval. But these constraints do not limit ambition. They define the path forward.

At Ipsen, we believe that progress depends on shared responsibility. It is built through long-term partnerships, continuous improvement, and a willingness to tackle the most complex parts of the challenge.
On World Environment Day, the message is clear: we want to reduce our impact and we act where it matters most. Building on what we have already achieved, we are accelerating action across our value chain.
By Christelle Huguet, EVP & Head of R&D, Ipsen
In biopharmaceutical research, not all challenges are equal. Some of the most meaningful opportunities lie in areas defined by complex biology, limited evidence, and patients who have waited far too long for effective treatment options.
Why we focus where others hesitate
At Ipsen, we have made a deliberate choice to focus on these areas of highest unmet need. They demand deeper scientific insight, greater adaptability, and long-term commitment, but they also offer the greatest potential to deliver truly transformational impact for patients.
High unmet need diseases often fall outside traditional development models, requiring new ways of thinking and working. Patient populations may be small or heterogeneous. Endpoints may be harder to define. Biology may still be unfolding. Yet this is precisely where innovation can be most powerful and where progress can change lives.
Turning scientific ambition into meaningful progress
Taking on these challenges means embracing uncertainty. It pushes us to ask better questions, to work more collaboratively, and to design smarter development programs.
We build deep understanding of disease mechanisms and continuously adapt as new insights emerge. This allows us to develop programs that are both scientifically rigorous and closely aligned with patient needs. By applying this discipline, we can take thoughtful, intentional risks, prioritizing programs with first‑ or best‑in‑class potential and creating new possibilities where few existed before.
Raising expectations through impact
By focusing on the hardest problems, we seek not only to deliver new therapies, but also to help redefine how progress is measured in complex and rare populations. In doing so, we aim to raise expectations, demonstrating that rigorous, patient‑centred innovation is possible even in the most challenging settings.
Choosing the hardest problems is not the simplest path. But for patients with the greatest unmet needs, it is the path that matters most and one we are proud to take in pursuit of scientific progress.
By Sandra Silvestri, for Ipsen.com
In today’s rapidly evolving treatment landscape the approval of a medicine is no longer the finish line; it’s just the beginning. Real world evidence (RWE), post-hoc analyses and structured Phase IV studies all offer new opportunities for scientific learning. At Ipsen, we’re committed to pursuing the unanswered questions, generating robust evidence that goes beyond medicine development, which provides broader long-term evidence-based perspectives for the communities we serve.
The expanding role of RWE
Clinical trials answer essential scientific questions, but by design, they can never fully capture the reality of people’s everyday lives—the diversity of experiences, the complexity of conditions, and the unpredictability of care journeys.
RWE helps bridge that gap. It reflects what really matters to people: how well a medicine works over time, how tolerable it is day-to-day, and how it fits into the realities of living with a condition. By investing in robust RWE, we are deepening our understanding of patients’ needs, capturing the genuine impact of our medicines and ensuring they continue to deliver meaningful benefits for the people who rely on them.
Post-hoc analyses and Phase IV trials: deepening insight where it matters most
Post hoc analyses and Phase IV studies, conducted after a medicine is approved, play an equally important role. Directed by the diversity of patients and the often-varied care pathways, we explore assumptions to generate insights in the context of real-world challenges. How do our medicines impact patients with certain comorbidities or receiving other medications? What other markers may be clinically relevant to investigate? How has the treatment landscape and in turn, treatment guidelines evolved? By taking a focused approach, we’re designing analyses to answer the most pressing topics for clinicians and patients, enriching understanding and supporting more individualized care.
A future defined by continuous learning
Real world insights combined with the depth of Phase IV trials and post-hoc analyses, offer a powerful, multidimensional understanding of a medicine’s impact. For Ipsen, data generation post approval is not an add on, it is a commitment. A commitment to truly understand the impact of our medicines, not just for the many but for the unique individuals who deserve answers.
The future of patient care will depend on this continuous learning mindset. Approval is a milestone worth celebrating, but it is not the end. Every real-world insight, every long-term outcome, every post-approval analysis brings us closer to optimizing care for the people who need it most. Our goal is clear: to ensure that scientific progress does not stop at approval but accelerates through it so that every patient receives not only an innovative medicine, but one that evolves with evidence, precision and purpose.
A diagnosis is supposed to be the end of the uncertainty. For some families, it is also the moment they realize there is nothing more medicine can offer. Loubna Ouriaghli, Head of Government Affairs & Policy at Ipsen, witnessed that moment with her own mother who was diagnosed with a rare disease. She saw the symptoms appear one by one, the years it took to finally have an answer and the silence that followed when that answer came with no treatment.
That feeling of powerlessness became a turning point. Rather than resigning herself to it, Loubna chose to transform it into commitment with her expertise, her position, her skills so that other families, other children like her, would not have to face the same situation. As Loubna is growing a family, this personal perspective is further deepening her commitment to improving outcomes for future generations.
.
Since then, she approaches her role with a clarity that goes beyond professional responsibility. Government Affairs and Policy may not seem like an obvious place for that kind of purpose. It is the work of negotiations, frameworks, and political relationships. But it is also precisely where the gap between a treatment existing and a patient actually receiving it gets decided. Rare disease patients don’t just need treatments to exist, they need those treatments to be recognized, reimbursed, and made accessible. For someone with Loubna’s background, that work carries a weight that goes beyond the professional.
She knows the road is long, and that no single person can change the system alone. But for someone who has seen what is at stake, that is not a reason to step back. It is reason enough to act.
What we heard at the EASL Congress 2026!
We are back from the European Association for the Study of the Liver (EASL) Congress 2026, Europe’s largest event dedicated to liver health where doctors, scientists, allied health professionals, patients, and industry partners come together to share knowledge, highlight scientific breakthroughs, and build meaningful connections.
Our presence at EASL Congress 2026 reflected our continued commitment to driving progress in rare liver diseases. Through six scientific presentations – including three late‑breaking posters – expert‑led symposia and Meet the Expert sessions, we contributed to the advancement of science and supporting meaningful conversations about improvements in patient care.
At Ipsen, rare liver diseases are a core focus of our portfolio. We apply our scientific expertise across five rare cholestatic liver diseases: primary biliary cholangitis (PBC), progressive familial intrahepatic cholestasis (PFIC), Alagille syndrome (ALGS), biliary atresia (BA), and primary sclerosing cholangitis (PSC).
Through our clinical development programs, we aim to advance scientific understanding of rare liver diseases and their impact on the people who live with them. As these conditions can present in unpredictable ways, personalized management is essential, not only to control disease progression but also to address the symptoms that matter most to people living with a rare liver disease.
Outcomes improve when people living with a rare liver disease understand their condition, and when they are active partners in ongoing goal setting and reviews with their healthcare teams.
A collaborative, holistic approach means bringing together patients, carers, and multidisciplinary professionals, who listen to each other and understand the impact of diseases, including life impacting symptoms such as fatigue and pruritus. This will ensure that management plans reflect clinical priorities while fully accounting for the realities of living with a rare liver disease.
Ipsen’s Hugo Gomes da Silva has been sharing exclusive behind-the-scenes insights from EASL Congress 2026
Reporting live from EASL Congress 2026, Hugo Gomes da Silva, SVP Global Medical Affairs (Rare Disease) captured key moments and standout highlights shaping this year’s meeting. New episodes will be uploaded daily throughout the congress:
Learning more about PBC and Fatigue
An exciting development at this year’s EASL Congress 2026, is the increased recognition of the need to assess and actively manage fatigue in PBC due to its significant burden.
Fatigue in PBC is far more than just being tired; it is a distinct, debilitating clinical symptom that fundamentally alters a person’s quality of life. It is a profound, persistent lack of energy that forces difficult daily choices, impacting everything from physical movement and cognitive function to emotional and social well-being. By affecting even the simplest tasks and personal interactions, PBC-related fatigue necessitates a dedicated approach to care.
Discover more about the reality of living with PBC fatigue here.
Bringing the patient voice to Barcelona
Ipsen is shaping the dialogue around PBC, advocating for proactive management that addresses both disease progression and the debilitating symptoms impacting patients’ daily lives.
During EASL Congress 2026, the streets of Barcelona featured the powerful testimonials of Gill, Jo, and Wendy from the This is My PBC campaign. By bringing their lived experiences directly to the medical community, these stories served as a vital reminder to attending HCPs that effective care requires a dual focus on both clinical disease control and symptom management.

Seeing our data spark discussion and recognizing its potential to influence real‑world care reinforced an important truth: our science is driven by purpose
Olivia Brown, EVP, Head of Neuroscience
There are moments that stay with you. Not because of a single slide or headline, but because you can feel something changing. Expectations shift. Standards rise. A new chapter begins.
That is what SCALE represented this year: it signalled the directions where aesthetic medicines is heading. Aesthetic medicine is evolving beyond surface‑level outcomes. Physicians increasingly seek treatments grounded in evidence, developed with intention, and delivered to consistently high standards.
For Ipsen, this moment also matters. We are not new to aesthetics. For decades, our development in neuroscience has spanned both therapeutic and aesthetic indications, supported by deep experience in complex biologics and high‑quality manufacturing. Our medicines are approved for aesthetic use in more than 80 countries worldwide, through our direct presence and trusted partners.
What differentiates companies in this field is their approach and execution.
Aesthetics addresses individual patient needs. Behind every treatment decision is a human story: a change in life, confidence, identity, or sense of self. People are looking for a quality of life, not solely an aesthetic enhancement.
That is why the future of aesthetics will be shaped by a more intentional standard, one that respects the individual, values consistency and predictability, and recognizes the responsibility that comes with developing, manufacturing and delivering these treatments.
At Ipsen, we follow the science and let data guide our decisions. This principle extends beyond research. It means refusing shortcuts, designing with purpose and committing to quality at every step – from development through to manufacturing.
SCALE marked that shift. Not just a congress, but a point in time that reflects the direction the aesthetics industry is taking and the standards the community expects. It was also a milestone for Ipsen, as our role in aesthetics became more visible.
For me personally, this moment carries added significance. Since joining Ipsen I found a sharper awareness of the moments that shape not just careers, but responsibility for the standards we set and the trust we earn. SCALE was one of those moments.
A reminder that aesthetics is entering a more considered, more intentional era – one where scientific rigour, quality and standards matter as much as outcomes. And one where how we innovate is just as important as what we create.

Ipsen’s 2025 Integrated Annual report report is now available on Ipsen.com. This essential reference document brings together key information and insights into the major developments in 2025 and what lies ahead.
This year’s report offers a clear view of how our strategy, ‘Focus. Together. For patients & society,’ has translated into progress through our business performance, pipeline advancement, culture, sustainability and, above all, impact for patients, since its launch in 2020.
“Guided by our strategy, we have embarked on exciting new partnerships, expanded our pipeline and portfolio and shared promising data from our clinical trials. All of this broadened the range of options for patients living with difficult-to-treat conditions in oncology, rare disease and neuroscience,” says David Loew, CEO.
Read David Loew’s Q&A for additional insights on the progress achieved and the priorities ahead, or download the full report.
“All medicines have side effects, but it’s just about trying to minimize the impact through providing the information patients and clinicians need or finding other ways to mitigate that impact.” — Jacintha Sivarajah
With over 15 years of experience in the pharmaceutical industry, Jacintha now leads safety oversight for developing medicines. “My role is not only to ensure a proactive safety strategy but also, together with my Global Patient Safety colleagues, to provide robust safety surveillance for all patients enrolled across the whole program.”
She also sees safety science as a way of maintaining the crucial balance between risk and benefit. “What I do is help to ensure that whatever we do—whether it’s in our clinical trial programs or looking after the safety reporting when people report side effects—we assess that against the benefits and risks of treatments,” she says. “We aim to ensure that the benefit–risk balance remains in favor of the patients taking them.”
That balance requires vigilance and evolving tools. “Signal detection is a really important part of what we do in safety,” she explains. “You’re looking to see whether there are patterns emerging in side effects and whether that is associated with our medicine or not.” For Jacintha, the science of safety is about putting patients first.
“Ultimately, we all work to focus on the well-being of patients,” she says. “That’s what drives us all.”
Christelle Huguet, EVP Head of R&D
Ipsen is not new to aesthetics. For more than three decades, we have been advancing neuroscience innovations across therapeutic and aesthetic indications, building deep expertise in complex biologics, rigorous clinical development and high‑quality manufacturing. Our approved medicines are approved for aesthetic use in more than 80 countries worldwide, through our direct presence and trusted partners.
This heritage matters because the field of aesthetics is evolving, and its future will be shaped by science and data.
At Ipsen, innovation has always followed understanding. We build on what we know, learn from data and apply the same scientific discipline across therapeutic and aesthetic medicine. That approach has allowed us to translate decades of neuroscience research into solutions designed to meet rising expectations around quality, consistency and clinical robustness.
Today, expectations are changing. Patients, consumers and their treating physicians want treatments grounded in evidence, designed with intention and developed to consistently high standards. That requires more than incremental iteration, it demands deep biological insight, rigorous development pathways and confidence in the science behind every decision.
This conviction is reflected in how we invest. Today, 15% of Ipsen’s total R&D investment is dedicated to aesthetic indications.* We are advancing multiple Phase II and Phase III studies across aesthetic uses, reinforcing our vision to remain the engine of meaningful innovation in aesthetics.
Science‑led innovation also depends on the ability to translate discovery into reality. Ipsen’s manufacturing facilities are equipped with capabilities designed for the next generation of complex biologic treatments. They reflect long‑term investment in quality, reproducibility and manufacturing excellence – capabilities that matter when science moves from hypothesis to patient.
Aesthetics is advancing rapidly. But progress that lasts is built on evidence, experience and execution.
At Ipsen, we continue to shape the future of aesthetics through evidence based research, data driven decisions and building on decades of neuroscience expertise to deliver innovation.
*Reflects Ipsen R&D spend projected for 2026. Internal figure, February 2026.
We have delivered a strong start to 2026, reporting total sales growth of 22.6% at constant exchange rates in the first quarter. The performance reflects solid execution across our core therapeutic areas and continued demand for our differentiated medicines.
The Q1 results underscore our focus on delivering meaningful innovation for patients while driving sustainable, long‑term growth.
Consistent growth across all core therapeutic areas
Our first‑quarter performance was supported by balanced growth across Oncology, Rare Disease, and Neuroscience, driven by the strength and diversification of our portfolio.
- Oncology sales increased by 13.0%, with by continued strong demand across key brands and markets.
- Rare Disease recorded exceptional growth of 125.4%, reflecting successful commercialization efforts and increasing patient access to Ipsen’s rare disease treatments.
- Neuroscience sales grew by 18.5%, supported by sustained underlying demand and effective commercial execution.
This performance demonstrates our ability to deliver growth across multiple therapeutic areas addressing high unmet medical needs.
Well positioned for the year ahead
We’re starting 2026 with momentum and confirmed our 2026 full-year guidance. Looking ahead, we remain focused on advancing our pipeline, supporting launches, and reinforcing our position in Oncology, Rare Disease, and Neuroscience.
* All figures at constant exchange rates.